Abstract
Review Article
A Resurgence of the Idea of Hypertriglyceridemia and Lower Serum (HDL-C) as Predictive Factors for Insulin Resistance (IR) & Type 2 Diabetes Mellitus Development: A Narrative Review
Kulvinder Kochar Kaur*
Published: 24 July, 2025 | Volume 9 - Issue 1 | Pages: 001-012
Read Full Article HTML DOI: 10.29328/journal.niogb.1001022 Cite this Article Read Full Article PDF
References
- Obesity and overweight. Updated 9 June 2021. Accessed 10 January 2024. Available from: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight
- Pluta W, Dudzinska W, Lubkowska A. Metabolic obesity in people with normal weight (MONW) – review of diagnostic criteria. Indian J Inf Public Health. 2022;19(2):395–402. Available from: https://doi.org/10.3390/ijerph19020624
- Kaur KK, Allahbadia GN, Singh M. An update on microRNA's and metabolic regulation with future therapeutic potentials regarding diagnosis and treatment of obesity, metabolic syndrome and other related disorders. J Health Med Informat. 2015;6(2). Available from: https://www.hilarispublisher.com/archive/jhmi-volume-6-issue-2-year-2015.html
- Kaur KK, Allahbadia GN, Singh M. Chronically elevated triglycerides as a result of high fat palatable diet resulting in a vicious cycle on reinforcing reward and dopamine signaling: a possible cause for the obesity epidemic worldwide in the food environment available – a comprehensive review. J Endocrinol. 2019;3(2):1–20. Available from: https://medwinpublishers.com/article-description.php?artId=4605
- Kaur KK, Allahbadia GN, Singh M. An update on aetiopathogenesis and management of obesity. Obes Control Therap. 2016;3(1):1–17. Available from: http://dx.doi.org/10.15226/2374-8354/2/2/00123
- Kaur KK, Allahbadia GN, Singh M. Existing and prospective pathways for intervention in treatment of obesity in a novel way – a review. MOJ Drug Des Develop Ther. 2018;2(3):95–105. Available from: https://medcraveonline.com/MOJDDT/MOJDDT-02-00035.pdf
- Kaur KK, Allahbadia GN, Singh M. Importance of simultaneous treatment of obesity and diabetes mellitus: a sequelae to the understanding of diabesity – a review. Obes Res Open J. 2019;6(1):1–10. Available from: http://dx.doi.org/10.17140/OROJ-6-136
- Kaur KK, Allahbadia GN, Singh M. Have probiotics and synbiotics passed the test of time to be implemented in the management of obesity and related metabolic disorders: comprehensive review. Adv Obes Weight Manag Control. 2019;9(1):21–28. Available from: https://doi.org/10.15406/aowmc.2019.09.00269
- Kaur KK, Allahbadia GN, Singh M. The association of dietary fatty acids and gut microbiota alterations in the development of neuropsychiatric diseases: a systematic review. Obes Res Open J. 2020;7(1):19–45. Available from: http://dx.doi.org/10.17140/OROJ-7-143
- Kaur KK, Allahbadia GN, Singh M. Therapeutic potential and epigenetic alterations of plant phytochemicals (as epi-drugs) for the treatment of type 2 diabetes mellitus: a systematic review. Adv Obes Weight Manag Control. 2021;11(6):195–206. Available from: https://medcraveonline.com/AOWMC/AOWMC-11-00355.pdf
- Kaur KK, Allahbadia GN, Singh M. Potential role of epigenetic modulation in prevention or therapy for diabetic kidney disease – still a dream or a reality: a systematic review. J Diab Nephro Diab Mgmt. 2021;1:1–26. Available from: https://www.researchgate.net/publication/351632704_Potential_Role_of_Epigenetic_Modulation_in_Prevention_or_Therapy_for_Diabetic_Kidney_Disease-Still_a_Dream_or_a_Reality_-A_Systematic_Review_Diabetic_Nephropathy_and_Diabetes_Management_Introduction-D
- Kaur KK, Allahbadia GN, Singh M. Role of adipocyte impairment in heart failure induction in subjects that are obese along with prediabetes and overt diabetes mellitus: a systematic review. Int J Cardiol Card. 2021;2(2):1–21. Available from: https://unisciencepub.com/abstract/role-of-adipocyte-impairment-in-heart-failure-induction-in-subjects-that-are-obese-along-with-prediabetes-and-overt-diabetes-mellitus-a-systematic-review/
- Kaur KK, Allahbadia GN, Singh M. Are we any closer to unraveling the mechanism of interactions among susceptibility genes towards type 1 diabetes, gut microbiota along with environmental factors, specifically early diet patterns: a systematic review. J Endocrinol Surg Endocrinol. 2021;2(1):10–34. Available from: https://www.researchgate.net/publication/349915934_Are_we_Any_Close_to_Unraveling_the_Mechanism_of_Interactions_Among_Susceptibility_Genes_Towards_Type_1_Diabetes_Gut_Microbiota_Along_with_Environmental_Factors_Specifically_Early_Diet_Patterns_-A_Syst
- Kaur KK, Allahbadia GN, Singh M. Diabetic cardiomyopathy: an update on its pathophysiology with specific emphasis on epigenetics modifications besides treatment: a systematic review. BOHR Int J Curr Res Diabetes Prev Med. 2022;1(1):1–16. Available from: http://dx.doi.org/10.54646/bijrdpm.001
- Kaur KK, Allahbadia GN, Singh M. De novo lipogenesis inhibitors: as the other innovative agents for therapy of metabolic diseases (obesity, NAFLD/NASH, CVD). Adv Obes Weight Manag Control. 2022;12(3):78–93. Available from: https://medcraveonline.com/AOWMC/AOWMC-12-00367.pdf
- Kaur KK, Allahbadia GN, Singh M. Identification of women of polycystic ovary syndrome (PCOS) at risk for generation of metabolic disease in normal weight PCOS: is there a role of flutamide? A short communication. CPQ Med. 2023;14(2):1–5. Available from: https://www.researchgate.net/publication/367497075_Identification_of_Women_of_Polycystic_Ovary_Syndrome_PCOS_At_Risk_for_Generation_of_Metabolic_Disease_in_Normal_Weight_PCOS_Is_There_a_Role_of_Flutamide-A_Short_Communication
- Kaur KK, Allahbadia GN, Singh M. Very low calorie ketogenic diet (VLCKD) possesses further advantageous actions over Mediterranean diet in terms of better enhancement of gut microbiota in type 2 diabetes mellitus patients: a short communication. J Diab Metab Disord. 2023;10(1):48–54. Available from: https://doi.org/10.15406/jdmdc.2023.10.00252
- Kaur KK, Allahbadia GN, Singh M. Inventing the immunomodulatory action of food plants with utilization of metabolomics: a narrative review. Acta Sci Nutr Health. 2023;7(10):21–36. Available from: https://actascientific.com/ASNH/pdf/ASNH-07-1308.pdf
- Kaur KK, Allahbadia GN, Singh M. The significance of gut microbiota controlling epigenetic modifications in obesity with its therapeutic impact: a narrative review. Adv Obes Weight Manag Control. 2025;15(1):1–10. Available from: https://www.researchgate.net/profile/Kulvinder-Kochar-Kaur/publication/389465035_AOWMC-15-00413gm-obes_-epigen-The_significance_of_gut_microbiota_controlling_of_epigenetic_modifications_in_obesity_with_its_therapeutic_impact_a_narrative_review/links/67c30cda8311ce680c791a3b/AOWMC-15-00413gm-obes-epigen-The-significance-of-gut-microbiota-controlling-of-epigenetic-modifications-in-obesity-with-its-therapeutic-impact-a-narrative-review.pdf
- Kaur KK, Allahbadia GN, Singh M. An update on targeting epigenetic modifications in obesity correlated diseases, for instance cancers, type 2 diabetes, and cardiovascular diseases (CVD) for their avoidance along with therapy: a narrative review. J Clin Med Images. Forthcoming 2025.
- Himsworth H. Diabetes mellitus: its differentiation into insulin sensitive and insulin insensitive types. Lancet. 1936;227:127–30. Available from: https://doi.org/10.1093/ije/dyt203
- Reaven GM, Lerner RL, Stern MP, Farquhar JW. Role of insulin in endogenous hypertriglyceridemia. J Clin Invest. 1967;46:1756–67. Available from: https://doi.org/10.1172/jci105666
- Ford S Jr, Bozian RC, Knowles HC Jr. Interactions of obesity, glucose, and insulin levels in hypertriglyceridemia. Am J Clin Nutr. 1968;21:904–10. Available from: https://doi.org/10.1093/ajcn/21.9.904
- Steiner G, Vranic M. Hyperinsulinemia and hypertriglyceridemia: a vicious cycle with atherogenic potential. Int J Obes. 1982;6(Suppl 1):117–24. Available from: https://pubmed.ncbi.nlm.nih.gov/6749716/
- Reaven GM. Banting lecture 1988: role of insulin resistance in human disease. Diabetes. 1988;37:1595–607. Available from: https://doi.org/10.2337/diab.37.12.1595
- Reaven G. Insulin resistance and coronary heart disease in nondiabetic individuals. Arterioscler Thromb Vasc Biol. 2012;32:1754–9. Available from: https://doi.org/10.1161/atvbaha.111.241885
- von Eckardstein A, Sibler RA. Possible contributions of lipoproteins and cholesterol to the pathogenesis of type 2 diabetes mellitus. Curr Opin Lipidol. 2011;22:26–32. Available from: https://doi.org/10.1097/mol.0b013e3283412279
- Drew BG, Rye KA, Duffy SJ, Barter P, Kingwell BA. The emerging role of HDL in glucose metabolism. Nat Rev Endocrinol. 2012;8:237–45. Available from: https://doi.org/10.1038/nrendo.2011.235
- Li N, Fu J, Koonen DP, Kuivenhoven JA, Snieder A, Hofker MH. Are hypertriglyceridemia and low HDL causal factors in the development of insulin resistance? Atherosclerosis. 2014;233:130–8. Available from: https://doi.org/10.1016/j.atherosclerosis.2013.12.013
- Lee Y, Hirose H, Ohneda M, Johnson JH, McGarry JD, Unger RH. Beta-cell lipotoxicity in the pathogenesis of non-insulin-dependent diabetes mellitus of obese rats: impairment in adipocyte-beta-cell relationships. Proc Natl Acad Sci U S A. 1994;91:10878–82. Available from: https://doi.org/10.1073/pnas.91.23.10878
- Unger RH. Lipotoxicity in the pathogenesis of obesity-dependent NIDDM: genetic and clinical implications. Diabetes. 1995;44:863–70. Available from: https://doi.org/10.2337/diab.44.8.863
- Unger RH, Scherer PE. Gluttony, sloth and the metabolic syndrome: a roadmap to lipotoxicity. Trends Endocrinol Metab. 2010;21(6):345–52. Available from: https://doi.org/10.1016/j.tem.2010.01.009
- Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive summary of the third report of the National Cholesterol Education Program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III). JAMA. 2001;285:2486–97. Available from: https://doi.org/10.1001/jama.285.19.2486
- Després JP, Lemieux I. Abdominal obesity and metabolic syndrome. Nature. 2006;444:881–7. Available from: https://doi.org/10.1038/nature05488
- Després JP, Lemieux I, Bergeron J, Pibarot P, Mathieu P, Larose E, et al. Abdominal obesity and the metabolic syndrome: contribution to global cardiometabolic risk. Arterioscler Thromb Vasc Biol. 2008;28:1039–49. Available from: https://doi.org/10.1161/atvbaha.107.159228
- Lean ME, Han TS, Morrison CE. Waist circumference as a measure for indicating the need for weight management. BMJ. 1995;311:158–61. Available from: https://doi.org/10.1136/bmj.311.6998.158
- Després JP. Excess visceral adipose tissue/ectopic fat: the missing link in the obesity paradox? J Am Coll Cardiol. 2011;57:1887–9. Available from: https://doi.org/10.1016/j.jacc.2010.10.063
- Ross R, Neeland IJ, Yamashita S, Shai I, Seidell J, Magni P, et al. Waist circumference as a vital sign in clinical practice: a consensus statement from the IAS and ICCR Working Group on Visceral Obesity. Nat Rev Endocrinol. 2020;16:177–89. Available from: https://doi.org/10.1038/s41574-019-0310-7
- Gami AS, Witt BJ, Howard DE, Erwin PJ, Gami LA, Somers VK, Montori VM. Metabolic syndrome and risk of incident cardiovascular events and death: a systematic review and meta-analysis of longitudinal studies. J Am Coll Cardiol. 2007;49:403–14. Available from: https://doi.org/10.1016/j.jacc.2006.09.032
- Mottillo S, Filion KB, Genest J, Joseph L, Pilote L, Poirier P, et al. The metabolic syndrome and cardiovascular risk: a systematic review and meta-analysis. J Am Coll Cardiol. 2010;56:1113–32. Available from: https://doi.org/10.1016/j.jacc.2010.05.034
- Ford ES, Li C, Sattar N. Metabolic syndrome and incident diabetes: current state of the evidence. Diabetes Care. 2008;31:1898–904. Available from: https://doi.org/10.2337/dc08-0423
- Alberti KG, Eckel RH, Grundy SM, Zimmet PZ, Cleeman JI, Donato KA, et al. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation. 2009;120:1640–5. Available from: https://doi.org/10.1161/circulationaha.109.192644
- Tokunaga K, Matsuzawa Y, Ishikawa K, Tarui S. A novel technique for the determination of body fat by computed tomography. Int J Obes. 1983;7:437–45. Available from: https://pubmed.ncbi.nlm.nih.gov/6642855/
- Ferland M, Després JP, Tremblay A, Pinault S, Nadeau A, Moorjani S, et al. Assessment of adipose tissue distribution by computed axial tomography in obese women: association with body density and anthropometric measurements. Br J Nutr. 1989;61:139–48. Available from: https://doi.org/10.1079/bjn19890104
- Ross R, Freeman J, Hudson R, Janssen I. Abdominal obesity, muscle composition, and insulin resistance in premenopausal women. J Clin Endocrinol Metab. 2002;87:5044–51. Available from: https://doi.org/10.1210/jc.2002-020570
- Matsuzawa Y, Funahashi T, Nakamura T. The concept of metabolic syndrome: contribution of visceral fat accumulation and its molecular mechanism. J Atheroscler Thromb. 2011;18:629–39. Available from: https://doi.org/10.5551/jat.7922
- Neeland IJ, Ross R, Després JP, Matsuzawa Y, Yamashita S, Shai I, et al. Visceral and ectopic fat, atherosclerosis, and cardiometabolic disease: a position statement. Lancet Diabetes Endocrinol. 2019;7:715–25. Available from: https://doi.org/10.1016/s2213-8587(19)30084-1
- Stefan N, Schick F, Haring HU. Causes, characteristics, and consequences of metabolically unhealthy normal weight in humans. Cell Metab. 2017;26:292–300. Available from: https://doi.org/10.1016/j.cmet.2017.07.008
- Nazare JA, Smith JD, Borel AL, Haffner SM, Balkau B, Ross R, et al. Ethnic influences on the relations between abdominal subcutaneous and visceral adiposity, liver fat, and cardiometabolic risk profile: the International Study of Prediction of Intra-Abdominal Adiposity and Its Relationship With Cardiometabolic Risk/Intra-Abdominal Adiposity. Am J Clin Nutr. 2012;96:714–26. Available from: https://doi.org/10.3945/ajcn.112.035758
- Després JP. The Reaven syndrome: a tribute to a giant. Nat Rev Endocrinol. 2018;14:319–20. Available from: https://doi.org/10.1038/s41574-018-0013-5
- Kurooka N, Eguchi J, Wada J. Role of glycosylphosphatidylinositol-anchored high-density lipoprotein binding protein 1 in hypertriglyceridemia and diabetes. J Diabetes Investig. 2023;14(10):1148–56. Available from: https://doi.org/10.1111/jdi.14056
- Schaefer EJ, Anthanont P, Diffenderfer MR, Polisecki E, Asztalos BF. Diagnosis and treatment of high density lipoprotein deficiency. Prog Cardiovasc Dis. 2016;59(2):97–106. Available from: https://doi.org/10.1016/j.pcad.2016.08.006
- Krishnappa M, Patil K, Parmar K, Trivedi P, Mody N, Shah C, et al. Effect of saroglitazar 2 mg and 4 mg on glycemic control, lipid profile, and cardiovascular disease risk in patients with type 2 diabetes mellitus: a 56-week, randomized, double blind, phase 3 study (PRESS XII study). Cardiovasc Diabetol. 2020;19(1):93. Available from: https://doi.org/10.1186/s12933-020-01073-w
- Simental-Mendia LE, Rodriguez-Moran M, Guerrero-Romero F. The product of fasting glucose and triglycerides is a surrogate for identifying insulin resistance in apparently healthy subjects. Metab Syndr Relat Disord. 2008;6(4):299–304. Available from: https://doi.org/10.1089/met.2008.0034
- Guerrero-Romero F, Simental-Mendia LE, Gonzalez-Ortiz M, Martinez-Abundis E, Ramos-Zavala MG, Hernandez-Gonzalez SO, et al. The product of triglycerides and glucose, a simple measure of insulin sensitivity. Comparison with the euglycemic-hyperinsulinemic clamp. J Clin Endocrinol Metab. 2010;95(7):3347–51. Available from: https://doi.org/10.1210/jc.2010-0288
- Vasques AC, Novaes FS, de Oliveira Mda S, Souza JR, Yamanaka A, Pareja JC, et al. TyG index performs better than HOMA in a Brazilian population: a hyperglycemic clamp validated study. Diabetes Res Clin Pract. 2011;93(3):e98–100. Available from: https://doi.org/10.1016/j.diabres.2011.05.030
- Lee SH, Kwon HS, Park YM, Ha HS, Jeong SH, Yang HK, et al. Predicting the development of diabetes using the product of triglycerides and glucose: the Chungju Metabolic Disease Cohort (CMC) study. PLoS One. 2014;9(2):e90430. Available from: https://doi.org/10.1371/journal.pone.0090430
- Placzkowska S, Pawlik-Sobecka L, Kokot I, Piwowar A. Indirect insulin resistance detection: current clinical trends and laboratory limitations. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2019;163(3):187–99. Available from: https://doi.org/10.5507/bp.2019.021
- Nabipoorashrafi SA, Seyedi SA, Rabizadeh S, Ebrahimi M, Ranjbar SA, Reyhan SK, et al. The accuracy of triglyceride-glucose (TyG) index for the screening of metabolic syndrome in adults: a systematic review and meta-analysis. Nutr Metab Cardiovasc Dis. 2022;32(12):2677–88. Available from: https://doi.org/10.1016/j.numecd.2022.07.024
- Lyu L, Wang X, Xu J, Liu Z, He Y, Zhu W, et al. Association between triglyceride glucose-body mass index and long-term adverse outcomes of heart failure patients with coronary heart disease. Cardiovasc Diabetol. 2024;23(1):162. Available from: https://link.springer.com/article/10.1186/s12933-024-02213-2
- Zheng S, Shi S, Ren X, Han T, Li Y, Chen Y, et al. Triglyceride-glucose-waist circumference, a novel and effective predictor of diabetes in first-degree relatives of type 2 diabetes patients: cross-sectional and prospective cohort study. J Transl Med. 2016;14(1):260. Available from: https://doi.org/10.1186/s12967-016-1020-8
- Mirr M, Braszak-Cymerman A, Ludziejewska A, Kręgielska-Narożna M, Bogdański P, Bryl W, et al. Serum asprosin correlates with indirect insulin resistance indices. Biomedicines. 2023;11(6):1568. Available from: https://doi.org/10.3390/biomedicines11061568
- Darshan An V, Rajput R, Meena, Mohini, Garg R, Saini S. Comparison of triglyceride glucose index and HbA1C as a marker of prediabetes - A preliminary study. Diabetes Metab Syndr. 2022;16(9):102605. Available from: https://doi.org/10.1016/j.dsx.2022.102605
- Er LK, Wu S, Chou HH, Hsu LA, Teng MS, Sun YC, et al. Triglyceride glucose-body mass index is a simple and clinically useful surrogate marker for insulin resistance in nondiabetic individuals. PLoS One. 2016;11(3):e0149731. Available from: https://doi.org/10.1371/journal.pone.0149731
- Dang K, Wang X, Hu J, Zhang Y, Cheng L, Qi X, et al. The association between triglyceride-glucose index and its combination with obesity indicators and cardiovascular disease: NHANES 2003–2018. Cardiovasc Diabetol. 2024;23(1):8. Available from: https://doi.org/10.1186/s12933-023-02115-9
- International Diabetes Federation (IDF). IDF Diabetes Atlas. 10th ed. Brussels: International Diabetes Federation; 2022.
- Gong R, Liu Y, Luo G, Liu W, Jin Z, Xu Z, et al. Associations of TG/HDL ratio with the risk of prediabetes and diabetes in Chinese adults: A Chinese Population Cohort Study based on open data. Int J Endocrinol. 2021;2021:9949579. Available from: https://doi.org/10.1155/2021/9949579
- Neuenschwander M, Ballon A, Weber KS, Norat T, Aune D, Schwingshackl L, et al. Role of diet in type 2 diabetes incidence: Umbrella review of meta-analyses of prospective observational studies. BMJ. 2019;366:l2368. Available from: https://doi.org/10.1136/bmj.l2368
- Wu Y, Ding Y, Tanaka Y, Zhang W. Risk factors contributing to type 2 diabetes and recent advances in the treatment and prevention. Int J Med Sci. 2014;11(11):1185–200. Available from: https://doi.org/10.7150/ijms.10001
- Tinajero MG, Malik VS. An update on the epidemiology of type 2 diabetes: A global perspective. Endocrinol Metab Clin North Am. 2021;50(3):337–55. Available from: https://doi.org/10.1016/j.ecl.2021.05.013
- World Obesity Federation. World Obesity Atlas 2022. World Obesity Federation; 2022. Available from: https://www.worldobesity.org/resources/resource-library/world-obesity-atlas-2022
- Qin Y, Qiao Y, Wang D, Li M, Yang Z, Li L, et al. Visceral adiposity index is positively associated with fasting plasma glucose: A cross-sectional study from National Health and Nutrition Examination Survey 2017–2020. BMC Public Health. 2023;23(1):313. Available from: https://link.springer.com/article/10.1186/s12889-023-15231-8
- Neeland IJ, Ross R, Després JP, Matsuzawa Y, Yamashita S, Shai I, et al. Visceral and ectopic fat, atherosclerosis, and cardiometabolic disease: A position statement. Lancet Diabetes Endocrinol. 2019;7(9):715–25. Available from: https://doi.org/10.1016/S2213-8587(19)30084-1
- Karastergiou K. The interplay between sex, ethnicity, and adipose tissue characteristics. Curr Obes Rep. 2015;4(2):269–78. Available from: https://link.springer.com/article/10.1007/s13679-015-0149-8
- Qin Z, Chen X, Sun J, Jiang L. The association between visceral adiposity index and decreased renal function: A population-based study. Front Nutr. 2023;10:1076301. Available from: https://doi.org/10.3389/fnut.2023.1076301
- Amato MC, Giordano C, Galia M, Criscimanna A, Vitabile S, Midiri M, et al. Visceral Adiposity Index: A reliable indicator of visceral fat function associated with cardiometabolic risk. Diabetes Care. 2010;33(4):920–2. Available from: https://doi.org/10.2337/dc09-1825
- Wei J, Liu X, Xue H, Wang Y, Shi Z. Comparisons of visceral adiposity index, body shape index, body mass index, and waist circumference and their associations with diabetes mellitus in adults. Nutrients. 2019;11(7):1580. Available from: https://doi.org/10.3390/nu11071580
- Zhang X, Sun Y, Li Y, Wang C, Wang Y, Dong M, et al. Association between visceral adiposity index and heart failure: A cross-sectional study. Clin Cardiol. 2023;46(3):310–9. Available from: https://doi.org/10.1002/clc.23976
- Wang J, Chen S, Zhao J, Liang J, Gao X, Gao Q, et al. Association between nutrient patterns and hyperuricemia: Mediation analysis involving obesity indicators in the NHANES. BMC Public Health. 2022;22(1):1981. Available from: https://link.springer.com/article/10.1186/s12889-022-14357-5
- Zhang Y, He Q, Zhang W, Xiong Y, Shen S, Yang J, et al. Non-linear associations between visceral adiposity index and cardiovascular and cerebrovascular diseases: Results from the NHANES (1999–2018). Front Cardiovasc Med. 2022;9:908020. Available from: https://doi.org/10.3389/fcvm.2022.908020
- Chen Q, Zhang Z, Luo N, Qi Y. Elevated visceral adiposity index is associated with increased stroke prevalence and earlier age at first stroke onset: Based on a national cross-sectional study. Front Endocrinol. 2023;13:1086936. Available from: https://doi.org/10.3389/fendo.2022.1086936
- Zhou H, Li T, Zhuang X, Yang J. The association between visceral adiposity index and risk of type 2 diabetes mellitus. Sci Rep. 2024;14:16634. Available from: https://www.nature.com/articles/s41598-024-67430-x
- Kaur KK, Allahbadia GN, Singh M. Impact of nutrigenomics on various metabolic disorders about lifestyle alteration. Austin J Nutr Food Sci. 2018;6(1):1100. Available from: https://d1wqtxts1xzle7.cloudfront.net/106284396/ajnfs_v6_id11002_corrected_NUTRIGENOMICS.pdf
- Kaur KK, Allahbadia GN, Singh M. An update on the therapeutic potential of herbal preparations with regards to molecular and biochemical mechanisms in the management of diabetes mellitus: a systematic review. World J Adv Healthc Res. 2022;6(3):825–35.
- Dama A, Shpati K, Daliu P, et al. Targeting metabolic diseases: The role of nutraceuticals in modulating oxidative stress and inflammation. Nutrients. 2024;16(4):507. Available from: https://doi.org/10.3390/nu16040507
- Kaur KK, Allahbadia GN, Singh M. A review of nutrient metabolism in obesity with special emphasis on fatty acid metabolism. BAOJ Food Sci Technol. 2017;1:001. Available from: https://www.researchgate.net/publication/313165171
- Kaur KK, Allahbadia GN, Singh M. Synthesis and functional significance of polyunsaturated fatty acids (PUFAs) in body. Acta Sci Nutr Health. 2018;2(4):43–50. Available from: https://www.researchgate.net/publication/324111487
- Kaur KK, Allahbadia GN, Singh M. The association of dietary fatty acids and gut microbiota alterations in the development of neuropsychiatric diseases: A systematic review. Obes Res Open J. 2020;7(1):19–45. Available from: http://dx.doi.org/10.17140/OROJ-7-143
- Elkanawati RY, Sumiwi SA, Levita J. Impact of lipids on insulin resistance: Insights from human and animal studies. Drug Des Devel Ther. 2024;18:3337–60. Available from: https://doi.org/10.2147/dddt.s468147
- Kaur KK, Allahbadia GN. An update on targeting ferroptosis for generating innovative strategies in treatment of diabetic kidney disease: a narrative review. Med Discoveries. 2025;4(6):1262. Available from: https://www.researchgate.net/publication/393361382
Figures:
Figure 1
Figure 2

Figure 3

Figure 4
Figure 5
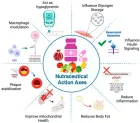
Figure 6
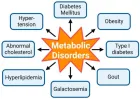
Figure 7
Similar Articles
-
Hypoglycemia by Ginseng in type 2 Diabetic Patient: Case ReportAngelo Michele Carella*,Teresa Marinelli,Armando Melfitano,Michele Di Pumpo,Matteo Conte,Angelo Benvenuto. Hypoglycemia by Ginseng in type 2 Diabetic Patient: Case Report . . 2017 doi: 10.29328/journal.hodms.1001001; 1: 001-006
-
Cardiac Autonomic Modulation in children and Preadolescents obeseMário Augusto Paschoal*. Cardiac Autonomic Modulation in children and Preadolescents obese. . 2017 doi: 10.29328/journal.hodms.1001002; 1: 007-009
-
The interaction between photonic technology and physical exercise: The action of low-level laser therapyAntonio Eduardo de Aquino Jr*,Fernanda Mansano Carbinatto. The interaction between photonic technology and physical exercise: The action of low-level laser therapy. . 2017 doi: 10.29328/journal.hodms.1001003; 1: 010-013
-
Behavioral factors of Abdominal Obesity and effects of lifestyle changes with Fiber AdequacyRoberto Carlos Burini*,Hugo Tadashi Kano,Mariana Santoro Nakagaki,Patricia Baston Frenhani,Katia Cristina Portero-McLellan. Behavioral factors of Abdominal Obesity and effects of lifestyle changes with Fiber Adequacy. . 2017 doi: 10.29328/journal.hodms.1001004; 1: 014-022
-
Study of body composition, physiological variables in Grade III obese submitted to arm ergometer testRenato Vidal Linhares*,Felipe Monnerat Marino Rosa,José Quaresma,João Regis Ivar Carneiro,Valéria Bender Braulio,Roberto Simão,Denise Pires de Carvalho,osé Fernandes Filho. Study of body composition, physiological variables in Grade III obese submitted to arm ergometer test . . 2017 doi: 10.29328/journal.hodms.1001005; 1: 023-029
-
Upper gut bacterial overgrowth is a potential mechanism for Glucose Malabsorption after Vertical Sleeve GastrectomyTimothy R Koch*,Timothy R Shope,Matthew E Sharbaugh. Upper gut bacterial overgrowth is a potential mechanism for Glucose Malabsorption after Vertical Sleeve Gastrectomy. . 2017 doi: 10.29328/journal.hodms.1001006; 1: 030-035
-
Body mass index in a group of security forces (policemen). Cross-sectional studyGuillermo Padrón Arredondo*. Body mass index in a group of security forces (policemen). Cross-sectional study. . 2018 doi: 10.29328/journal.niogb.1001007; 2: 001-004
-
Herbal approach for obesity managementPreeti Singh*. Herbal approach for obesity management. . 2018 doi: 10.29328/journal.niogb.1001008; 2: 005-016
-
Dietary and Lifestyles assessment among Obese Women in Gaza City, PalestineMarwan O Jalambo*,Basil Kanoa,Mohammed S Ellulu,Smaher Younis,Mueen El-Kariri. Dietary and Lifestyles assessment among Obese Women in Gaza City, Palestine. . 2018 doi: 10.29328/journal.niogb.1001009; 2: 017-025
-
Obesity-Treatment by drugsSahithi G*. Obesity-Treatment by drugs. . 2019 doi: 10.29328/journal.niogb.1001010; 3: 001-001
Recently Viewed
-
From Wallets to Waistlines: Exploring the Link between Family Income (Wage Distribution) and Obesity Indices in Pakistani PopulationsAtif Akbar,Waqas Ghulam Hussain*. From Wallets to Waistlines: Exploring the Link between Family Income (Wage Distribution) and Obesity Indices in Pakistani Populations. New Insights Obes Gene Beyond. 2026: doi: 10.29328/journal.niogb.1001024; 10: 001-014
-
Screening for Depressive Symptoms in Clinical and Nonclinical Youth: The Psychometric Properties of the Dutch Children’s Depression Inventory-2 (CDI-2)Denise HM Bodden*,Yvonne Stikkelbroek,Daan Creemers,Sanne PA Rasing,Elien De Caluwe,Caroline Braet. Screening for Depressive Symptoms in Clinical and Nonclinical Youth: The Psychometric Properties of the Dutch Children’s Depression Inventory-2 (CDI-2). Insights Depress Anxiety. 2025: doi: 10.29328/journal.ida.1001047; 9: 028-039
-
Knowledge, Attitude, and Practice of Healthcare Workers in Ekiti State, Nigeria on Prevention of Cervical CancerAde-Ojo Idowu Pius*, Okunola Temitope Omoladun, Olaogun Dominic Oluwole. Knowledge, Attitude, and Practice of Healthcare Workers in Ekiti State, Nigeria on Prevention of Cervical Cancer. Arch Cancer Sci Ther. 2024: doi: 10.29328/journal.acst.1001038; 8: 001-006
-
Oral Cancer Management is not just Treatment! But also, how early Pre-cancerous Lesions are Diagnosed & Treated!!Suresh Kishanrao*. Oral Cancer Management is not just Treatment! But also, how early Pre-cancerous Lesions are Diagnosed & Treated!!. Arch Cancer Sci Ther. 2024: doi: 10.29328/journal.acst.1001039; 8: 007-012
-
Breast Cancer in FemaleLorena Menditto*. Breast Cancer in Female. Arch Cancer Sci Ther. 2024: doi: 10.29328/journal.acst.1001040; 8: 013-018
Most Viewed
-
Physical Performance in the Overweight/Obesity Children Evaluation and RehabilitationCristina Popescu, Mircea-Sebastian Șerbănescu, Gigi Calin*, Magdalena Rodica Trăistaru. Physical Performance in the Overweight/Obesity Children Evaluation and Rehabilitation. Ann Clin Endocrinol Metabol. 2024 doi: 10.29328/journal.acem.1001030; 8: 004-012
-
Hypercalcaemic Crisis Associated with Hyperthyroidism: A Rare and Challenging PresentationKarthik Baburaj*, Priya Thottiyil Nair, Abeed Hussain, Vimal MV. Hypercalcaemic Crisis Associated with Hyperthyroidism: A Rare and Challenging Presentation. Ann Clin Endocrinol Metabol. 2024 doi: 10.29328/journal.acem.1001029; 8: 001-003
-
Effects of dietary supplementation on progression to type 2 diabetes in subjects with prediabetes: a single center randomized double-blind placebo-controlled trialSathit Niramitmahapanya*,Preeyapat Chattieng,Tiersidh Nasomphan,Korbtham Sathirakul. Effects of dietary supplementation on progression to type 2 diabetes in subjects with prediabetes: a single center randomized double-blind placebo-controlled trial. Ann Clin Endocrinol Metabol. 2023 doi: 10.29328/journal.acem.1001026; 7: 00-007
-
Exceptional cancer responders: A zone-to-goDaniel Gandia,Cecilia Suárez*. Exceptional cancer responders: A zone-to-go. Arch Cancer Sci Ther. 2023 doi: 10.29328/journal.acst.1001033; 7: 001-002
-
Ectopic adrenal tissue at the spermatic cordAbdallah Chaachou,Nizar Cherni,Wael Ferjaoui*,Mohamed Dridi,Samir Ghozzi. Ectopic adrenal tissue at the spermatic cord. J Clin Med Exp Images. 2022 doi: 10.29328/journal.jcmei.1001024; 6: 001-002

If you are already a member of our network and need to keep track of any developments regarding a question you have already submitted, click "take me to my Query."